Protocol - Myocardial Infarction
- Blood Pressure (Adult/Primary)
- Cigarette Smoking Status - Adolescent
- Cigarette Smoking Status - Adult
- Lipid Profile
- Weight - Measured Weight
- Weight - Self-Reported Weight
Description
This measure assesses if an individual has had a myocardial infarction (MI) through collection of personal history of disease, treatment and procedure histories, and medical record abstraction.
Specific Instructions
If the respondent answers "yes" to question 1 or question 2, the interviewer should complete the rest of the protocol. If the respondent answers "no" or "dont know," then the protocol is deemed complete.
Availability
This protocol is freely available; permission not required for use.
Protocol
1. Has a doctor ever told you that you had a myocardial infarction or heart attack?
[ ] 1 Yes
[ ] 0 No
[ ] 9 Dont Know
2. Have you had an outpatient or day surgery procedure to unblock blocked or narrowed blood vessels of the heart (called a PTCA, coronary angioplasty, stent, or atherectomy)?
[ ] 1 Yes
[ ] 0 No
Remainder of protocol to be abstracted from patients hospital medical records.
3. Was there an acute episode of pain, discomfort or tightness in the chest, left arm or jaw within 72 hours of the hospitalization or within 72 hours of the in-hospital event?
[ ] Yes
[ ] No
[ ] Unknown
4. Was the discomfort or pain diagnosed as having a non-cardiac origin?
[ ] Yes
[ ] No
[ ] Unknown
5. Were electrocardiograms (ECGs or EKGs) recorded?
[ ] Yes
[ ] No
[ ] Unknown
If "No" or "Unknown," skip to 6.
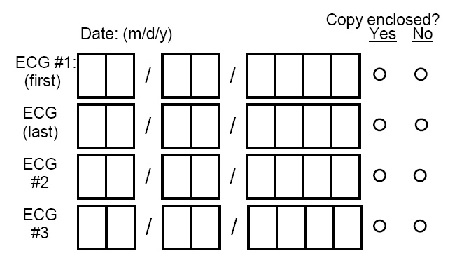
Record dates of ECGs and make two copies of FOUR ECG tracings as described below. Send one copy to the ECG Reading Center and attach one copy to this form:
-- If four or fewer tracings were made, include all tracings.
-- If more than four tracings were made, include:
1. First two codable tracings after admission (ECG#1-First and ECG#2)
2. Last codable tracing prior to discharge or death (discharge tracing) (ECG-Last)
3. Last codable tracing on day 3 (or the first tracing thereafter) following an admission or in-hospital event (ECG#3)
4. The next codable tracing after day 3
-- If the participant is readmitted (transferred) to the ICU/CCU because of a new episode of chest pain, the first codable tracing may be sent.
Serum Enzymes
6. Were any cardiac enzyme measurements performed during this admission?
[ ] Yes
[ ] No
If "No," skip to end.
7. Did the participant have any active liver disease (cirrhosis, hepatitis, liver cancer, etc.)?
[ ] Yes
[ ] No
If "Yes," specify:
_____________________________________________________
8. Is there any evidence of hemolytic disease during this hospitalization?
[ ] Yes
[ ] No
9. Is there any mention of the participant having either trauma, a _ procedure, or rhabdomyolysis within one week prior to the measurement of the cardiac enzymes?
[ ] Yes
[ ] No
[ ] Unknown
If "Yes," please specify type of trauma or procedure below.
Date m/d/y: __ / __ / __ __
Type of Trauma or procedure:
__________________________________________
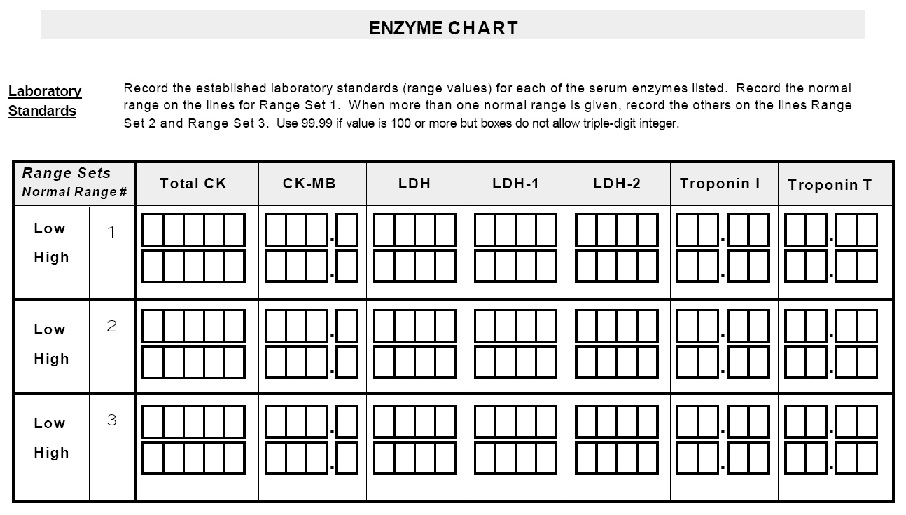
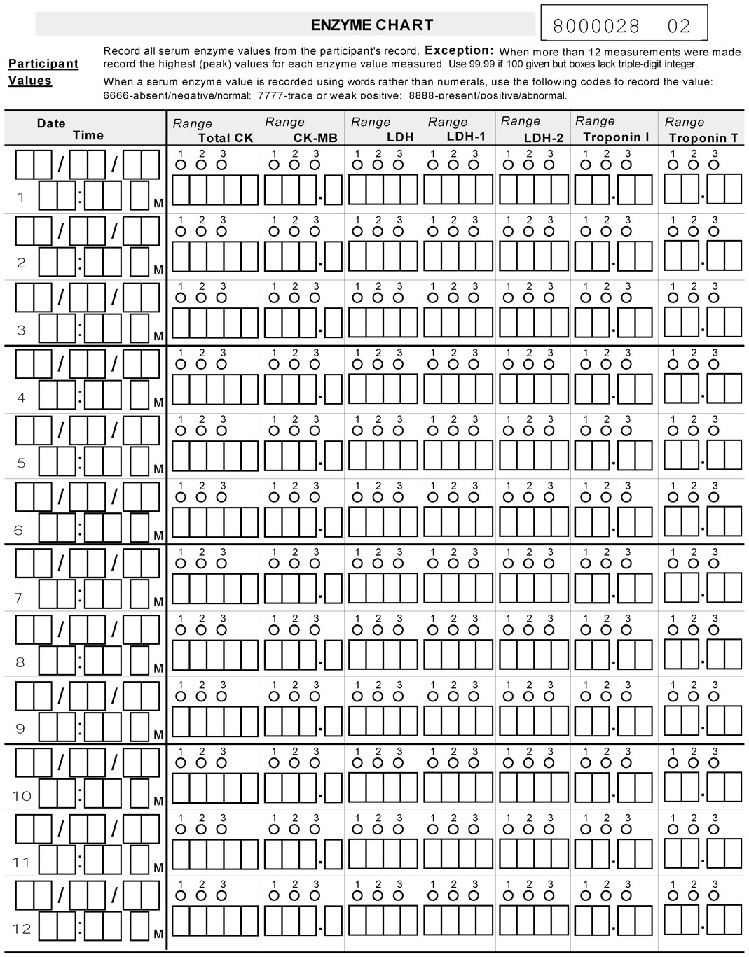
* Please complete ENZYME CHART. *
10. Myocardial infarction
[ ] Definite
[ ] Probable
[ ] No MI (skip to end)
If “Definite” or “Probable” enter date of MI (MM/DD/YYYY): __ __ / __ __ / __ __ __ __
Criteria
11. Chest Pain
[ ] Present
[ ] Absent
12. Cardiac Enzymes
[ ] Abnormal
[ ] Equivocal
[ ] Incomplete
[ ] Normal
13. ECG Serial Reading (pick one)
[ ] Evolution of Major Q-Wave
[ ] Evolution of ST-T Elevation with or without Q-Wave
[ ] New LBBB
[ ] Evolution of ST-Depression/Inversion alone
[ ] Evolution of Minor Q-Wave alone
[ ] Single ECG with Major Q-Wave
[ ] Single ECG with LBBB, described as new
[ ] Absent, Uncodable or Other ECG
14. Procedure-related:
[ ] Yes, cardiovascular
[ ] Yes, non-cardiovascular
[ ] No
Diagnostic Criteria:
Acute, Evolving or Recent MI
Either one of the following criteria satisfies the diagnosis for an acute, evolving or recent MI:
1. Typical rise and gradual fall (troponin) or more rapid rise and fall (CK-MB) of biochemical markers of myocardial necrosis with at least one of the following:
- ischemic symptoms;
- development of pathologic Q waves on the ECG;
- ECG changes indicative of ischemia (ST segment elevation or depression); or
- coronary artery intervention (e.g., coronary angioplasty).
2. Pathologic findings of an acute MI.
Established MI
Any one of the following criteria satisfies the diagnosis for established MI:
1. Development of new pathologic Q waves on serial ECGs. The patient may or may not remember previous symptoms. Biochemical markers of myocardial necrosis may have normalized, depending on the length of time that has passed since the infarct developed.
2. Pathologic findings of a healed or healing MI.
Personnel and Training Required
An interviewer who is trained to conduct personal interviews with individuals from the general population is required to complete the protocols from the Cardiovascular Health Study (CHS) and the Womens Health Initiative (WHI). The interviewer must be trained and found to be competent (i.e., tested by an expert) at the completion of personal interviews.* The interviewer should be trained to prompt respondents further if a "dont know" response is provided.
*There are multiple modes to administer this question (e.g., paper-and-pencil and computer-assisted interviews).
A trained and certified Hospital Record Abstractor is required to complete the protocol from the Multi-Ethnic Study of Atherosclerosis (MESA). This person should be trained to perform medical record and chart abstraction using various hospital records.
Equipment Needs
None
Requirements
| Requirement Category | Required |
|---|---|
| Major equipment | No |
| Specialized training | Yes |
| Specialized requirements for biospecimen collection | No |
| Average time of greater than 15 minutes in an unaffected individual | No |
Mode of Administration
Interviewer-administered question, Medical record abstraction
Lifestage
Adult
Participants
Cardiovascular Health Study (CHS): >65 years old
Womens Health Initiative (WHI): women ages 50–79 years old*
Multi-Ethnic Study of Atherosclerosis (MESA): ages 45–85 years old
*While this questionnaire was used in a womens study, the Cardiovascular Working Group deems it appropriate to use with men.
Selection Rationale
A combination of four protocols was chosen to obtain sufficient data to assess whether a person had a myocardial infarction. Obtaining a personal history of myocardial infarction and treatment procedures, in combination with medical record abstraction, will provide a high level of specificity and reliability.
Language
Chinese, English, Other languages available at source
Standards
| Standard | Name | ID | Source |
|---|---|---|---|
| Logical Observation Identifiers Names and Codes (LOINC) | Myocardial infarct proto | 62397-5 | LOINC |
| Human Phenotype Ontology | Myocardial infarction | HP:0001658 | HPO |
| caDSR Form | PhenX PX040801 - Myocardial Infarction | 5838015 | caDSR Form |
Derived Variables
None
Process and Review
Not applicable.
Protocol Name from Source
Cardiovascular Health Study (CHS), Baseline Medical History Questionnaire & Womens Health Initiative (WHI), Medical History Update
Source
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute. Cardiovascular haHealth Study (CHS). Baseline Medical History Questionnaire. Page 1. Question 1 (source for question 1).
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute. Womens Health Initiative (WHI). Form 33D—Medical History Update. Version 4. Question 3.3 (source for question 2).
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute. Multi-Ethnic Study of Atherosclerosis (MESA). Hospital Abstraction: Cardiac/Peripheral Vascular Disease (PVD) Form. Questions 15, 17, 27–31 (source for questions 3–9).
U.S. Department of Health and Human Services. National Institutes of Health. National Heart, Lung and Blood Institute. Multi-Ethnic Study of Atherosclerosis (MESA). Cardiac Review Form. Question 1 (source for questions 10–14 ).
Alpert, J. S., Thygesen, K., Antman, E., & Bassand, J. P. (2000). Myocardial infarction redefined—A consensus document of the Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. Journal of the American College of Cardiology, 36(3), 959–969 (source for Diagnostic Criteria).
General References
Ives, D. G., Fitzpatrick, A. L., Bild, D. E., Psaty, B. M., Kuller, L. H., Crowley, P. M., Cruise, R. G., & Theroux, S. (1995). Surveillance and ascertainment of cardiovascular events. The Cardiovascular Health Study. Annals of Epidemiology, 5, 278–285.
Psaty, B. M., Kuller, L. H., Bild, D., Burke, G. L., Kittner, S. J., Mittelmark, M., Price, T. R., Rautaharju, P. M., & Robbins, J. (1995). Methods of assessing prevalent cardiovascular disease in the Cardiovascular Health Study. Annals of Epidemiology, 5(4), 270–277.
Protocol ID
40801
Variables
Export Variables| Variable Name | Variable ID | Variable Description | dbGaP Mapping | |
|---|---|---|---|---|
| PX040801_Cardiac_Enzyme | ||||
| PX040801120000 | Cardiac Enzymes | N/A | ||
| PX040801_Chest_Discomfort | ||||
| PX040801030000 | Was there an acute episode of pain, more | N/A | ||
| PX040801_Chest_Discomfort_Non_Cardiac | ||||
| PX040801040000 | Was the discomfort or pain diagnosed as more | N/A | ||
| PX040801_Chest_Pain | ||||
| PX040801110000 | Chest Pain | N/A | ||
| PX040801_ECG_Serial_Reading | ||||
| PX040801130000 | ECG Serial Reading (pick one) | N/A | ||
| PX040801_Electrocardiogram | ||||
| PX040801050000 | Were electrocardiograms (ECGs or EKGs) recorded? | N/A | ||
| PX040801_Enzyme_Chart_Date | ||||
| PX040801090700 | Record all serum enzyme values Date (mm/dd/yy) | N/A | ||
| PX040801_Enzyme_Chart_Enzyme | ||||
| PX040801090300 | Record all serum enzyme values Serum enzyme | N/A | ||
| PX040801_Enzyme_Chart_Range | ||||
| PX040801090400 | Record all serum enzyme values Normal Range | N/A | ||
| PX040801_Enzyme_Chart_Time | ||||
| PX040801090800 | Record all serum enzyme values Time (hh:mm am/pm) | N/A | ||
| PX040801_Enzyme_Chart_Value_High | ||||
| PX040801090600 | Record all serum enzyme values Highest more | N/A | ||
| PX040801_Enzyme_Chart_Value_Low | ||||
| PX040801090500 | Record all serum enzyme values Lowest enzyme value | N/A | ||
| PX040801_Ever_Have_Myocardial_Infarction | ||||
| PX040801010000 | Has a doctor ever told you that you had a more | N/A | ||
| PX040801_Ever_Have_Stent | ||||
| PX040801020000 | Have you had an outpatient or day surgery more | N/A | ||
| PX040801_Evidence_Hemolytic_Disease | ||||
| PX040801080000 | Is there any evidence of hemolytic disease more | N/A | ||
| PX040801_Liver_Disease | ||||
| PX040801070000 | Did the participant have any active liver more | N/A | ||
| PX040801_Liver_Disease_Specify | ||||
| PX040801070100 | Did the participant have any active liver more | N/A | ||
| PX040801_Myocardial_Infarction | ||||
| PX040801100000 | Myocardial infarction | N/A | ||
| PX040801_Myocardial_Infarction_Date | ||||
| PX040801100100 | Myocardial infarction: If "Definite" or more | N/A | ||
| PX040801_Procedure_Related | ||||
| PX040801140000 | Procedure-related | N/A | ||
| PX040801_Serum_Enzyme | ||||
| PX040801060000 | Were any cardiac enzyme measurements more | N/A | ||
| PX040801_Trauma_Before_Enzyme_Measurement | ||||
| PX040801090000 | Is there any mention of the participant more | N/A | ||
| PX040801_Trauma_Before_Enzyme_Measurement_Date | ||||
| PX040801090100 | Is there any mention of the participant more | N/A | ||
| PX040801_Trauma_Before_Enzyme_Measurement_Type | ||||
| PX040801090200 | Is there any mention of the participant more | N/A | ||